2,4-Dinitrotoluene Chemistry
Molecular structure of 2,4-Dinitrotoluene (CAS NO.121-14-2) is:
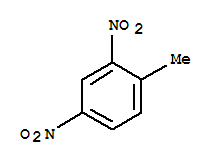
Product Name: 2,4-Dinitrotoluene
CAS Registry Number: 121-14-2
IUPAC Name: 1-Methyl-2,4-dinitrobenzene
Molecular Weight: 182.13354 [g/mol]
Molecular Formula: C7H6N2O4
XLogP3: 2
H-Bond Donor: 0
H-Bond Acceptor: 4
EINECS: 204-450-0
Melting Point: 67-70 ��C(lit.)
Surface Tension: 57.2 dyne/cm
Density: 1.407 g/cm3
Flash Point: 146.9 ��C
Enthalpy of Vaporization: 51.83 kJ/mol
Boiling Point: 299.9 ��C at 760 mmHg
Vapour Pressure: 0.00207 mmHg at 25 ��C
Water Solubility: 0.3 g/L (20 ��C)
Stability: Stable. Incompatible with oxidizing agents, reducing agents, strong bases.
Product Categories: Intermediates of Dyes and Pigments; Aromatic Hydrocarbons (substituted) & Derivatives; Alpha Sort; DAlphabetic; DID - DIN; DID - DINEnvironmental Standards; Method 8330Analytical Standards; Solid WasteMore...Close...; 8000 Series Solidwaste Methods; Analytical Standards; AromaticsAlphabetic; Chemical Class; D; Environmental Standards; ExplosivesEPA; Nitro CompoundsChromatography; Volatiles/ Semivolatiles; Nitro Compounds; Nitrogen Compounds; Organic Building Blocks; AromaticsVolatiles/ Semivolatiles; DID - DINAnalytical Standards
2,4-Dinitrotoluene Uses
2,4-Dinitrotoluene (CAS NO.121-14-2) is used in the production of toluene diisocyanate which is used to produce flexible polyurethane foams. It is hydrogenated to produce toluenediamine, which in turn is phosgenated to form toluene diisocyanate. Other uses include the explosives industry. also it used as material in organic synthesis, dyestuff and explosive.
2,4-Dinitrotoluene Production
The dinitrotoluenes [CH3C6H3(NO2)2] are manufactured by the nitration of toluene that gives a mixture containing 65 to 80% of the 2,4-dinitro derivative and 20 to 35% of the 2,6-dinitro compound.
2C6H5CH3 + 4HNO3 + 4H+ �� 2,4 (NO2)2C6H3CH3
+ 2,6(NO2)2C6H3CH3 + 4H2O
If the pure 2,4-compound is required, mononitration of toluene followed by separation of pure,p-nitrotoluene from the ortho isomer, and then further nitration of,p-nitrotoluene gives the pure 2,4-dinitro isomer.
2,4-Dinitrotoluene Toxicity Data With Reference
| Organism |
Test Type |
Route |
Reported Dose (Normalized Dose) |
Effect |
Source |
| cat |
LDLo |
subcutaneous |
25mg/kg (25mg/kg) |
|
U.S. Public Health Service, Public Health Bulletin. Vol. 271, Pg. 110, 1941. |
| guinea pig |
LD50 |
skin |
> 1gm/kg (1000mg/kg) |
|
National Technical Information Service. Vol. OTS0572393, |
| mouse |
LD50 |
oral |
790mg/kg (790mg/kg) |
|
Gigiena Truda i Professional'nye Zabolevaniya. Labor Hygiene and Occupational Diseases. Vol. 25(8), Pg. 50, 1981. |
| rat |
LD50 |
oral |
268mg/kg (268mg/kg) |
|
National Technical Information Service. Vol. PB214-270, |
2,4-Dinitrotoluene Consensus Reports
NCI Carcinogenesis Bioassay (feed); No Evidence: mouse NCITR* National Cancer Institute Carcinogenesis Technical Report Series. (Bethesda, MD 20014) No. NCI-CG-TR-54 ,1978. ; Some Evidence: rat NCITR* National Cancer Institute Carcinogenesis Technical Report Series. (Bethesda, MD 20014) No. NCI-CG-TR-54 ,1978. . Reported in EPA TSCA Inventory.
2,4-Dinitrotoluene Safety Profile
suspected carcinogen with experimental carcinogenic and neoplastigenic data. poison by ingestion and subcutaneous routes. experimental reproductive effects. a skin irritant. mutation data reported. an irritant and an allergen. can cause anemia, methemoglobinemia, cyanosis, and liver damage. combustible when exposed to heat or flame; can react with oxidizing materials. to fight fire, use water spray or mist, dry chemical. decomposes when heated to 250��c. there are instances of explosion during manufacture or storage. mixture with nitric acid is a high explosive. mixture with sodium carbonate can decompose with significant pressure increase at 210��c. mixtures with other alkalies may have the same effect. ignites on contact with sodium oxide. when heated to decomposition it emits toxic fumes of nox.
hazard codes: toxict, dangerousn, flammablef
risk statements: 45-23/24/25-48/22-51/53-62-68-39/23/24/25-11
r45:may cause cancer.
r23/24/25:toxic by inhalation, in contact with skin and if swallowed.
r48/22:harmful: danger of serious damage to health by prolonged exposure if swallowed.
r51/53:toxic to aquatic organisms, may cause long-term adverse effects in the aquatic environment.
r62:risk of impaired fertility.
r68:possible risk of irreversible effects.
r39/23/24/25:danger of very serious irreversible effects and toxic by inhalation, in contact with skin and if swallowed.
r11:highly flammable.
safety statements: 53-45-61-36/37
s53:avoid exposure - obtain special instructions before use.
s45:in case of accident or if you feel unwell, seek medical advice immediately (show the label whenever possible.)
s61:avoid release to the environment. refer to special instructions / safety data sheets.
s36/37:wear suitable protective clothing and gloves.
ridadr: un 3454 6.1/pg 2
wgk germany: 3
rtecs: xt1575000
hazardclass: 6.1
packinggroup: ii
2,4-Dinitrotoluene Standards and Recommendations
OSHA PEL: TWA 1.5 mg/m3 (skin)
NIOSH REL: (Dinitrotoluene) Reduce to lowest level
2,4-Dinitrotoluene Analytical Methods
For occupational chemical analysis use OSHA: #44.
2,4-Dinitrotoluene Specification
2,4-Dinitrotoluene , its cas register number is 121-14-2. It also can be called 1-Methyl-2,4-dinitrobenzene ; 2,4-DNT ; 2,4-Dinitro-1-methylbenzene; 2,4-Dinitromethylbenzene ; 2,4-Dinitrotoluol ; 4-Methyl-1,3-dinitrobenzene ; AI3-15342 ; CCRIS 268 ; HSDB 1144 ; NCI-C01865 ; NSC 7194 ; RCRA waste number U105 ; Toluene, 2,4-dinitro- ; Benzene, 1-methyl-2,4-dinitro- ; Toluene, 2,4-dinitro- ; Dinitrotoluene . It is a solidifies if allowed to cool.
THz spectroscopic investigation of 2,4-dinitrotoluene
We have investigated the terahertz (THz) spectrum of 2,4-DNT by using Fourier transform infrared spectroscopy in the 0.2-19.5 THz region. We also examined low-frequency intermolecular or phonon modes between 0.2 and 1.8 THz via THz time-domain spectroscopy. The extracted absorption coefficient and refractive index of an intermolecular band at 1.08 THz are ~110 cm -1 and 1.67, respectively. Density functional theory (DFT) was applied to obtain structure and vibrational frequencies of 2,4-DNT. The calculated results are in agreement with the experimental data. Observed vibrational frequencies have been interpreted using DFT. Two intermolecular or phonon modes were identified at 1.08 and 2.52 THz.
Degradation of 2,4-dinitrotoluene by persulfate activated with iron sulfides
Of the many oxidants studied to date for subsurface remediation, persulfate (S2O82?) is particularly attractive for its stability under ambient conditions and high reactivity upon activation. In this study we evaluated a new approach to activate persulfate, namely using pyrite (FeS2) or iron sulfide (FeS), with 2,4-dinitrotoluene (DNT) as a model contaminant. Pyrite was able to activate persulfate to degrade DNT. However, DNT degradation was rapidly halted presumably due to the low water solubility of pyrite and the presence of trace dissolved oxygen. In contrast, FeS was effective in activating persulfate to continuously and completely degrade DNT. Separate experiments with Fe2+ and HS? suggest that Fe2+, rather than HS?, released from FeS was involved in persulfate activation and was necessary for rapid degradation of DNT. Further experiments show that reduction products of DNT were oxidized much faster than DNT by FeS-activated persulfate. These findings and their implications are discussed in the context of subsurface remediation strategies for hydrocarbons and reducible contaminants such as nitroaromatic compounds.
Degradation of 2,4-dinitrotoluene by persulfate activated with zero-valent iron
The oxidation of 2,4-dinitrotoluene (DNT) by persulfate (S2O82?) activated with zero-valent iron (Feo) was studied through a series of batch experiments. The mechanism for Feo activation was investigated by comparing with Fe2+, and the effects of persulfate-to-iron ratio and pre-reduction on DNT oxidation were examined. DNT was stable in the presence of persulfate and transformed only when Feo was added. Most DNT was degraded oxidatively by Feo-activated persulfate, whereas direct reduction of DNT by Feo was unimportant. The rate of DNT degradation increased with higher Feo dose, presumably due to increasing activation of persulfate by Feo and Fe2+. In contrast to the Feo-persulfate system, where complete oxidation DNT was achieved, only �� 20% of DNT was degraded and the reaction was terminated rapidly when Feo was replaced with equimolar Fe2+. This indicates that Feo is more effective than Fe2+ as activating agent and potentially more suitable for environmental applications. The reduction products of DNT were more rapidly oxidized by persulfate than DNT, suggesting that converting the nitro groups of NACs to amino groups prior to oxidation can greatly enhance their oxidation. This suggests that a sequential Feo reduction�Cpersulfate oxidation process may be an effective strategy to promote NAC degradation.
Degradation of 2,4-Dinitrotoluene by the Lignin-degrading Fungus Phanerochaete chrysosporium
Under ligninolytic conditions, the white rot basidiomycete Phanerochaete chrysosporium mineralizes 2,4-dinitrotoluene (I). The pathway for the degradation of I was elucidated by the characterization of fungal metabolites and oxidation products generated by lignin peroxidase (LiP), manganese peroxidase (MnP), and crude intracellular cell extracts. The multistep pathway involves the initial reduction of I to yield 2-amino-4-nitrotoluene (II). II is oxidized by MnP to yield 4-nitro-1,2-benzoquinone (XII) and methanol. XII is then reduced to 4-nitro-1,2-hydroquinone (V), and the latter is methylated to 1,2-dimethoxy-4-nitrobenzene (X). 4-Nitro-1,2-hydroquinone (V) is also oxidized by MnP to yield nitrite and 2-hydroxybenzoquinone, which is reduced to form 1,2,4-trihydroxybenzene (VII). 1,2-Dimethoxy-4-nitrobenzene (X) is oxidized by LiP to yield nitrite, methanol, and 2-methoxy-1,4-benzoquinone (VI), which is reduced to form 2-methoxy-1,4-hydroquinone (IX). The latter is oxidized by LiP and MnP to 4-hydroxy-1,2-benzoquinone, which is reduced to 1,2,4-trihydroxybenzene (VII). The key intermediate 1,2,4-trihydroxybenzene is ring cleaved by intracellular cell extracts to produce, after reduction, beta-ketoadipic acid. In this pathway, initial reduction of a nitroaromatic group generates the peroxidase substrate II. Oxidation of II releases methanol and generates 4-nitro-1,2-benzoquinone (XII), which is recycled by reduction and methylation reactions to regenerate intermediates which are in turn substrates for peroxidase-catalyzed oxidation leading to removal of the second nitro group. Thus, this unique pathway apparently results in the removal of both aromatic nitro groups before ring cleavage takes place.
Aerobic Degradation of Dinitrotoluenes and Pathway for Bacterial Degradation of 2,6-Dinitrotoluene
An oxidative pathway for the mineralization of 2,4-dinitrotoluene (2,4-DNT) by Burkholderia sp. strain DNT has been reported previously. We report here the isolation of additional strains with the ability to mineralize 2,4-DNT by the same pathway and the isolation and characterization of bacterial strains that mineralize 2,6-dinitrotoluene (2,6-DNT) by a different pathway.Burkholderia cepacia strain JS850 andHydrogenophaga palleronii strain JS863 grew on 2,6-DNT as the sole source of carbon and nitrogen. The initial steps in the pathway for degradation of 2,6-DNT were determined by simultaneous induction, enzyme assays, and identification of metabolites through mass spectroscopy and nuclear magnetic resonance. 2,6-DNT was converted to 3-methyl-4-nitrocatechol by a dioxygenation reaction accompanied by the release of nitrite. 3-Methyl-4-nitrocatechol was the substrate for extradiol ring cleavage yielding 2-hydroxy-5-nitro-6-oxohepta-2,4-dienoic acid, which was converted to 2-hydroxy-5-nitropenta-2,4-dienoic acid. 2,4-DNT-degrading strains also converted 2,6-DNT to 3-methyl-4-nitrocatechol but did not metabolize the 3-methyl-4-nitrocatechol. Although 2,6-DNT prevented the degradation of 2,4-DNT by 2,4-DNT-degrading strains, the effect was not the result of inhibition of 2,4-DNT dioxygenase by 2,6-DNT or of 4-methyl-5-nitrocatechol monooxygenase by 3-methyl-4-nitrocatechol.
Identification of Products Resulting from the Biological Reduction of 2,4,6Trinitrotoluene, 2,4Dinitrotoluene, and 2,6Dinitrotoluene by Pseudomonas sp
Pseudomonas sp. clone A is able to use 2,4,6-trinitrotoluene (TNT), 2,4-dinitrotoluene, and 2,6-dinitrotoluene as an N-source after the enzymatic removal of nitro groups from the aromatic ring. We have identified in culture supernatants a hydride?TNT Meisenheimer complex, which is an intermediate in the removal of the first nitro group from the TNT aromatic ring. It has been shown in vitro that the hydride?TNT Meisenheimer complex is transformed to 2,4-dinitrotoluene and an unidentified compound, the molecular mass of which is 235. As side metabolites, reduced TNT derivativeswhich cannot be used as an N-source by this bacteriumappeared in culture supernatants. 2-Hydroxylamino-4,6-dinitrotoluene, 4-hydroxylamino-2,6-dinitrotoluene, 4-amino-2,6-dinitrotoluene, 2-amino-4,6-dinitrotoluene, and 2,4-diamino-6-nitrotoluene were found and chemically characterized. Spontaneous condensation of partially reduced TNT forms leads to the production of azoxytoluenes, and a possible condensation mechanism is discussed. For novel compounds, 1H NMR, UV, and infrared data are provided. Therefore, Pseudomonas sp. clone A carries out two different initial reductive reactions in the metabolism of 2,4,6-trinitrotoluene; one process removes a nitro group from the aromatic ring and allows it to be used as an N-source, while the other gives rise to side products.
|









